Logistics 101: Basics of Freight Shipping

If you're responsible for finding a new freight carrier for your company and you’re new to logistics, you probably have a lot of questions. By familiarizing yourself with the basics of freight shipping, you’ll feel a lot more comfortable navigating quotes and contracts. Keep reading to learn what you need to know to get started.
It's More Than Calling for a Pick-Up
At first glance, the shipping process seems simple. You look up a couple of freight carriers, compare prices, pick the best rate, and hire someone to pick up your stuff, right? Well, you can do it that way, but the selection process involves a bit more due diligence if you want to get the best price and terms.Before you request price quotes, you need to know:
- What kind of shipment you have
- Your shipment’s weight class
- How your freight needs to be packaged
- Your budget, how you plan to pay, and company payment terms
- Where, exactly, the shipment will be picked up and dropped off
- If you need any special equipment or services; i.e., a lift gate
While this isn’t an exhaustive list of need-to-knows, it’s a good short list to get you started. When you shop carriers, it’s also always good to ask about bulk discounts, promotions, and early-bird price cuts.
Common Shipment Types
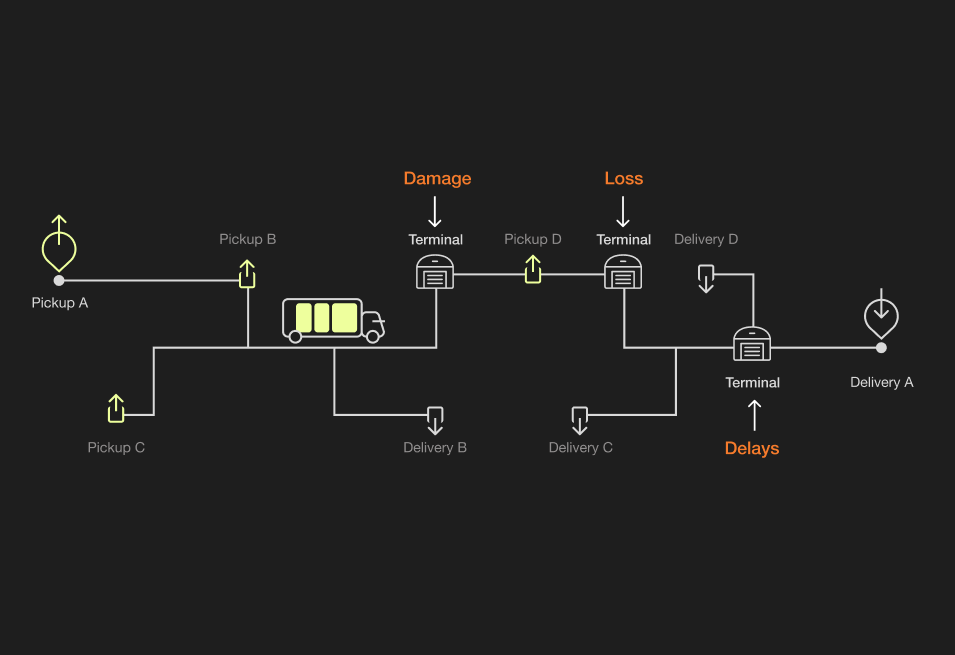
Step one in requesting a freight rate quote is knowing the size and weight of your shipment and identifying what type of carrier you need. LTL (less than load or less than truckload) and FTL (full truckload) shipments are the most common options, but PTL or shared truckloads also occur. Here are the basics of both common shipment options.Typically, an LTL shipment:
- Weighs less than 4,000 pounds
- Is shipped on skids or pallets; usually 1-6
- Goes on a truck with goods from other shippers and makes multiple stops or transfers
- Takes longer to get from origin to destination than an FTL shipment
In contrast, with FTL shipments:
- Freight can take up an entire semi-trailer
- Goods go directly from origin to destination
- The truck does not stop en-route or make transfers
- There’s less risk of damage, making FTL ideal for high-risk or fragile shipments
Once you determine what type of shipment you need, you can narrow down your carrier options.
Packing Shipments
Once you’ve determined what type of shipment you have, you need to figure out who is going to pack your shipment. You have three options:Option #1 - Your company can pack your freight at your warehouse or pickup location. This is usually the most cost-effective option. If you go this route, make sure you know what your carrier’s requirements are. Carriers can refuse shipments that aren’t packed correctly - and improper packing can cause items to get damaged in shipping.Option #2 - A third-party fulfillment company can pack your shipment for you. While this option is more convenient and eliminates the most risk of improper packing, it can come with a hefty fee.Option #3 - Your carrier can pack your freight (if packing is a service they offer). This may be the safest and easiest bet, but can also be quite expensive. However, the cost can be worth it for fragile, high-value shipments. Be sure to ask carriers if they offer any guarantees.
Who Pays the Bill and When is it Due?
Again, you typically have three options - and it depends on what you’re shipping, to whom, and why:Option #1 - Your company pays the bill after the freight is shippedOption #2 - The receiving party pays the bill when they get the shipmentOption #3 - A third party pays the bill at an agreed-upon time in the processThere are lots of different business arrangements and contract terms that can affect who pays for freight shipping and when the bill gets paid. Make sure you fully understand your company’s payment policies and the relationship between you (the shipper) and the person receiving your order. Then, verify that all payment terms outlined in your carrier contract are compliant.By understanding common freight terminology, asking the right questions, and familiarizing yourself with the shipping process, you can help your company secure the best service for the best price.